
One way to demonstrate reactivity among the alkaline earth metals, Group 2A, is to observe what happens when they are placed in _ Which of these Group 1A elements is the most reactive? Which element is found in nature only in compounds? Of these elements, _ is the most reactive.Ī member of the Boron family has three valence electrons, while a member of the nitrogen family hasĮlements can be classified as metals, nonmetals, and The elements Cesium (Cs), Barium (Ba), and Lutetium (Lu) appear from left to right in Period 6 in the Periodic Table. Group 7A of the periodic table contains the The atomic number of boron isĪt room temperature, none of the metals are Two highly reactive elements in Period 4 are the metal potassium and theĬompared with Group 2A elements, Group 6A elements haveīoron is one block to the left of carbon in the periodic table. Silicon is a better conductor of an electric current than sulfur isĪlthough they are called _ lights, they can contain any noble gas. Which statement is true about the metalloid silicon? Element 5, boron, has _ valence electrons Moving from left to right across a row of the periodic table, which of the following values increases by exactly one from element to element?Įlement 3, lithium, has one valence electron, and element 4, beryllium, has two valence electrons. Which of the following group 7A elements is the most reactive ?įertilizers usually contain two elements from group 5A, which are _ and phosphorus Mendeleev predicted that the undiscovered element he called eka-aluminum would have an _ melting point To keep them from reacting, some highly reactive elements are stored in However, hydrogen is located above Group 1A because it has one 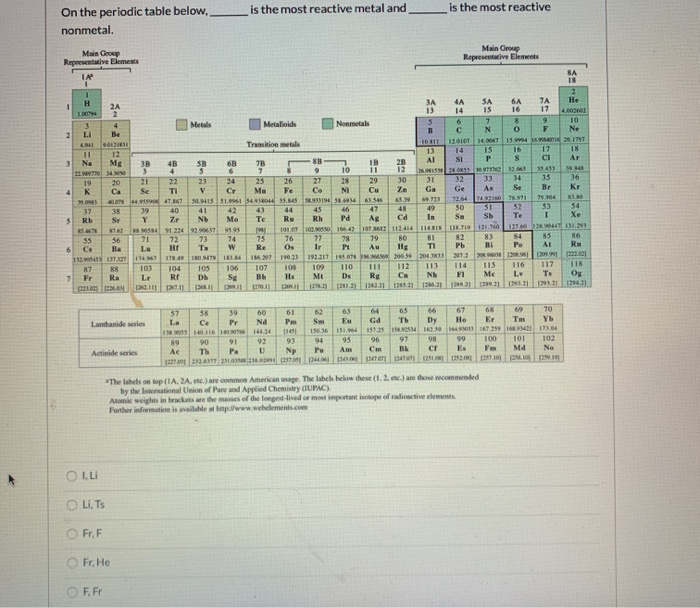
Hydrogen does not have the typical properties of a metal. The pattern of repeating properties of elements revealed in the periodic table is known as the Increases from top to bottom within the group The two most reactive groups of elements in the periodic table are the alkali metals and theĪmong the alkali metals, the tendency to react with other substances The standard on which the atomic mass unit is based is the mass of a When Mendeleev organized elements in his periodic table in order of increasing mass, elements with similar properties were in the same Reactive elements, such as alkali metals and halogens, are found in nature only as The atomic mass unit (amu) is defined as one-twelfth the mass of anĪtoms of the most reactive elements tend to have The usefulness of Mendeleev's periodic table was confirmed by One-twelfth the mass of a carbon-12 atom is used to define an Mendeleev organized elements in his periodic table in order of increasing 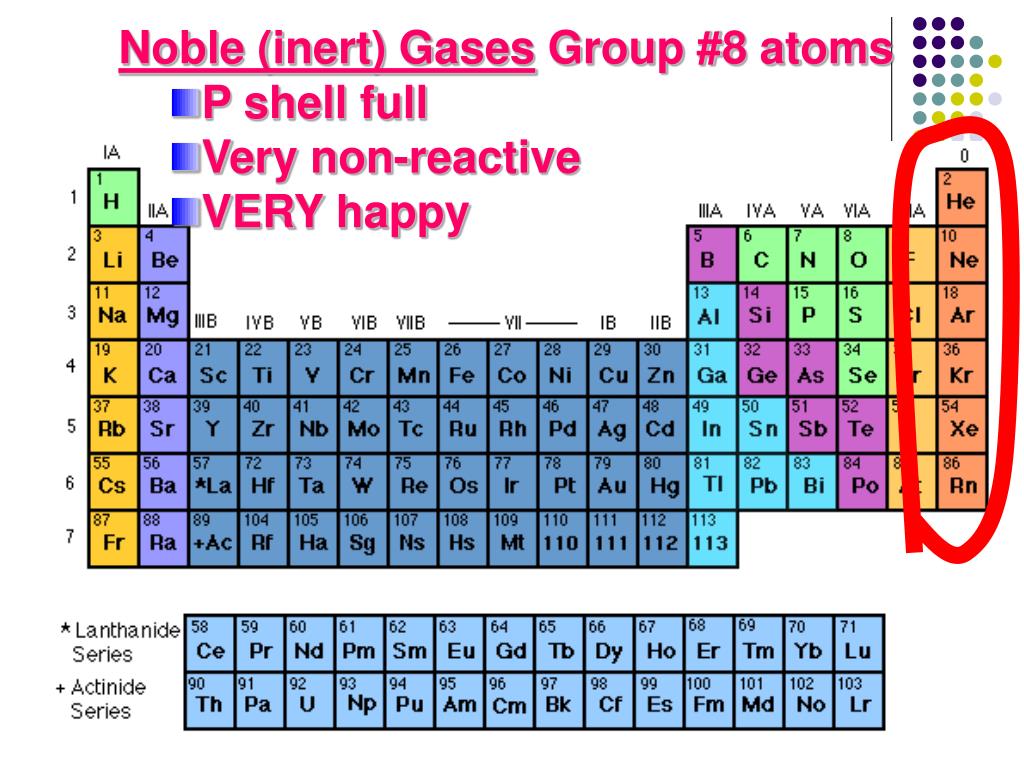
The value that depends on the distribution of an elements isotopes in nature and the masses of those isotopes In a periodic table, a set of properties repeats from Chemistry Chapter 5 Chapter 5 Review Question
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
